Overt Myelofibrosis (MF) is an end-stage subtype of BCR-ABL1 negative myeloproliferative neoplasms (MPN), which is characterized by progressive bone marrow (BM) fibrosis resulting in loss of normal hematopoietic support as well as inflammatory and vascular complications. While aberrant neoangiogenesis is known to participate in the pathogenesis of MF, Erba et al. (Am J Pathol, 2017) suggested the occurrence of endothelial-to-mesenchymal transition (EndMT) in BM and spleen of MF patients (pts). Given the relevance of endothelial cells (EC) for MF development and the occurrence of JAK2V617F-mutated EC in MPN pts, targeting of the vascular niche has emerged as a promising therapeutic strategy. To untangle the impact of the JAK2V617F mutation on human vasculogenesis, we leveraged high-purity and clonal patient-specific induced pluripotent stem cell (iPSC)-derived EC (iEC) harboring either the JAK2V617F mutation in hetero- (JAK2V617F HET) or homozygosis (JAK2V617F HOM). JAK2 WT and CRISPR-repaired isogenic JAK2 WT (both referred to as JAK2 WT) were used as controls.
In a first set of in vitro experiments, JAK2V617F mutated iEC showed a significantly increased proliferation rate in comparison to JAK2 WT iEC. We next evaluated the effect of selected drugs on iEC viability to assess their vascular toxicity. While treatment with 1 µM each of fedratinib, anagrelide or venetoclax led to a decrease in iEC viability (all **p ≤ .01), no significant alterations were observed with 1 µM ruxolitinib (RUX) or 1000 U/ml interferon-alpha (IFNa). Overall, no differences between the 3 genotypes were observed, pointing towards a genotype-independent mode of action. Given the favorable vascular toxicity profile of IFNa, as shown above, its anti-angiogenic potential was then assessed via endothelial tube formation assays. Pre-incubation with 1000 U/ml IFNa prior to seeding of the cells onto a reconstituted basement membrane matrix led to a significant inhibition of capillary-like structures formation in JAK2 WT and JAK2V617F HET or HOM iEC.
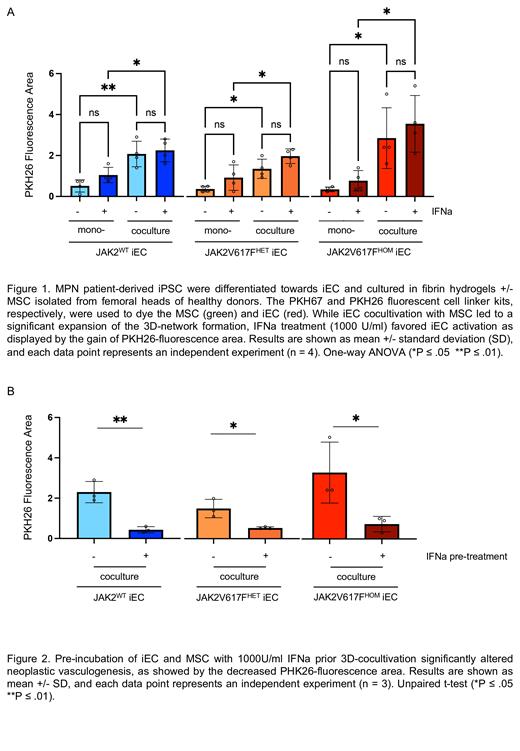
One of the limitations of 2D culture assays is the lack of 3D homotypic cellular organization, compromising its translational value. Therefore, we established an adaptive 3D fibrin-based model to recapitulate, in vitro, the critical EC-pericyte crosstalk required for formation and maturation of blood vessels. Recent studies highlighted the capacity of BM mesenchymal stem cells (MSC) to differentiate into pericytes. Therefore, we seeded JAK2 WT and JAK2V617F HETor HOM iEC with or without MSC in fibrin hydrogels. MSC and iEC were visualized using different PKH dyes as fluorescent cell linkers to enable live tracking of vascular tree formation. Although self-assembly of JAK2 WT and JAK2V617F HET or HOM iEC into vessel-like structures was observed in the generated monocultures, the presence of MSC in the cocultures led to a significant increase of the formed network area (Fig. 1), hence recapitulating the critical support of pericytes. While acute IFNa exposure did not alter vascular tree formation (Fig. 1), a separate pre-treatment of iEC and MSC prior to cocultivation, significantly impaired neoplastic vasculogenesis (Fig. 2). Interestingly, interactive microscopy image analysis revealed an increased MSC/iEC interaction in JAK2V617F HOM pre-treated cocultures (*p ≤ .05). Bulk RNA-sequencing of JAK2 WT and JAK2V617F HET or HOM iEC treated for 24h with 1000 U/ml showed a strong response to IFNa. Future analysis may unravel a JAK2V617F specific transcription signature. Moreover, we induced EndMT in our 3D iEC-MSC coculture model by treatment with pro-inflammatory cytokines, reflected by the loss of EC markers (e.g. Claudin 5) and upregulation of mesenchymal markers (e.g. aSMA) by immunofluorescence. We are currently evaluating the capacity of RUX vs IFNa to inhibit EndMT.
In summary, we show that IFNa impairs neoplastic vasculogenesis in a robust in vitro 3D coculture system that enables precise control of cellularity and genotype. Our model is a valuable tool to decipher the poorly investigated impact of IFNa on the BM microenvironment. In addition, its translational and scalable properties (e.g. by integrating primary or iPSC-derived hematopoietic cells) will allow us to study the impact of additional drugs on MPN-associated fibrotic transformation of the BM.
Disclosures
Caduc:AbbVie: Honoraria. Brümmendorf:Gilead: Consultancy, Speakers Bureau; Merck: Consultancy, Honoraria, Speakers Bureau; Novartis: Consultancy, Honoraria, Patents & Royalties, Research Funding, Speakers Bureau; Pfizer: Consultancy, Honoraria, Research Funding, Speakers Bureau; Janssen: Consultancy, Honoraria, Speakers Bureau. Koschmieder:Imago Bioscience: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Novartis, BMS/Celgene, Pfizer, Incyte, AOP Orphan, GSK, AbbVie, MPN Hub, Bedrock, iOMEDICO: Honoraria; Novartis: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support, Research Funding; Ariad: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; Pfizer, Incyte, Ariad, Novartis, AOP Pharma, BMS, Celgene, Geron, Janssen, CTI BioPharma, Roche, Bayer, GSK, Sierra Oncology, AbbVie, Protagonist, PharmaEssentia: Other: Advisory board; RWTH Aachen University: Patents & Royalties: BET inhibitor; AbbVie: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Janssen: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support, Research Funding; AOP Pharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support, Research Funding; Alexion, Novartis, Bristol Myers Squibb, Incyte, AOP Pharma, CTI BioPharma, Pfizer, Celgene, Janssen, Geron, Roche, AbbVie, GSK, Sierra Oncology, Kartos, Imago Biosciences, MSD, iOMEDICO: Other: Travel/accommodation support; Pfizer, Incyte, Ariad, Novartis, AOP Pharma, Bristol Myers Squibb, Celgene, Geron, Janssen, CTI BioPharma, Roche, Bayer, GSK, Protagonist, MPN Hub, Bedrock, PharmaEssentia: Consultancy; Incyte: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Bristol Meyers Squibb: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Squibb: Consultancy, Honoraria; CTI Biopharma: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Roche: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Pfizer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Geron, Janssen, AOP Pharma, Novartis: Research Funding; BMS/Celgene: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Protagonist: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; GSK: Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Bayer: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; PharmaEssentia: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees; RWTH Aachen University: Patents & Royalties: patent issued for a BET inhibitor; Sierra Oncology: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Karthos: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support; Geron: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: travel/accommodation support, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal